Biologics Contract Development Market Size, Share Report, Analysis, Trends, Growth 2032

Biologics Contract Development Market By Source (mammalian, microbial, and others), By Product (cell line development and process development), By Disease (oncology, immunological disorders, cardiovascular disorders, hematological disorders, and others) And By Region: - Global And Regional Industry Overview, Market Intelligence, Comprehensive Analysis, Historical Data, And Forecasts, 2024-2032
| Market Size in 2023 | Market Forecast in 2032 | Growth Rate (in %) | Base Year |
|---|---|---|---|
| USD 7.96 Billion | USD 16.18 Billion | CAGR at 8.20% | 2023 |
Description
Global Biologics Contract Development Market: Insights
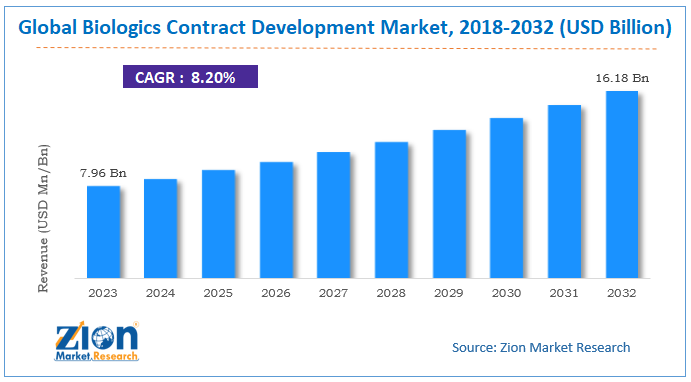
According to the report published by Zion Market Research, the global Biologics Contract Development Market size was valued at USD 7.96 Billion in 2023 and is predicted to reach USD 16.18 Billion by the end of 2032. The market is expected to grow with a CAGR of 8.20% during the forecast period. The report analyzes the global Biologics Contract Development Market’s growth drivers, restraints, and impact on demand during the forecast period. It will also help navigate and explore the arising opportunities in the Biologics Contract Development Market industry.
Global Biologics Contract Development Market: Overview
Biologics contract development is the comprehensive outsourcing service that manufactures biotechnology, pharmaceutical, and medical devices supporting biomaterials under the label of another company.
Global Biologics Contract Development Market: Growth Factors
Some of the significant factors that boost the global biologics contract development market are the surge in the adoption of advanced techniques for biological production, outsourcing the research & development tasks by pharma companies, and a well-suited environment for performing clinical trials. The surge in merger and collaboration activities increases the outsourcing of pharma and biopharma companies to expand in the developed and developing countries.
For instance, Thermo Fisher Scientific Inc. acquired Brammer Bio, a leader in contract development of cell and gene therapy, to empower its portfolio in gene therapy.
There has been a surge in diseases like cancer, neurology disorders, cardiovascular disorder, gynecological disorders, and ophthalmic disorders in recent years. The healthcare CDMOS emerged as a savior in patient care and many pharma leaders are making their contribution in inventing novel therapies for the efficient treatment of these diseases.
The increase in research and development investment for future advancements in technology fuels the market to grow significantly with high rates.
Biological contracts gradually shift to single-use bioreactors to facilitate the reduction in upfront costs and enhance manufacturing flexibility. Improved flexibility in manufacturing productivity increases the supply of biology and thus drives the global biologics contract development market.
Key Insights
- As per the analysis shared by our research analyst, the global Biologics Contract Development Market is estimated to grow annually at a CAGR of around 8.20% over the forecast period (2024-2032).
- In terms of revenue, the global Biologics Contract Development Market size was valued at around USD 7.96 Billion in 2023 and is projected to reach USD 16.18 Billion by 2032.
- Based on the source, the mammalian source segment accounted for the largest revenue share of 55.17%. This dominance is attributed to the ability of mammalian cells, such as Chinese Hamster Ovary (CHO) cell lines, to produce proteins with correct post-translational modifications, ensuring native protein structures. Approximately 70% of protein therapeutics are developed using CHO cells.
- Based on the product, the process development services held the largest revenue share of 74.5%. This category encompasses upstream and downstream process development, including cell line selection, culture medium optimization, and bioreactor system design. Companies like AGC Biologics and Bionova Scientific offer comprehensive upstream process development services tailored to client needs.
- Based on the disease, the oncology segment represented the largest revenue share of 49.8% in 2023. The increasing incidence of cancer and the growing investment in novel biologic therapies for cancer treatment have propelled this segment's dominance. Biologic therapies are pivotal in repairing, stimulating, or enhancing the immune response against cancer cells.
- Based on the region, the North America dominated the market, accounting for a revenue share of 43.16%. This leadership is due to advanced healthcare infrastructure, significant R&D investments, and the presence of major biopharmaceutical companies. The U.S., in particular, is projected to lead the global market in terms of revenue by 2030.
Biologics Contract Development Market: Dynamics
Key Growth Drivers
The biologics contract development market is experiencing significant growth driven by the increasing demand for biologic drugs, including monoclonal antibodies, vaccines, and cell and gene therapies. Pharmaceutical and biotechnology companies are increasingly outsourcing biologics development to contract development organizations (CDOs) to reduce operational costs and accelerate time-to-market. Additionally, advancements in bioprocessing technologies and the rise of personalized medicine have boosted the demand for specialized biologics development services. Favorable regulatory policies supporting the approval of biologics and the growing investments in biotechnology research further contribute to the market's expansion.
Restraints
Despite its growth, the biologics contract development market faces restraints such as the high costs associated with biologics development and manufacturing. Establishing and maintaining specialized facilities and hiring skilled professionals involve significant financial investments. Moreover, the complexity of biologics production increases the risk of regulatory non-compliance, leading to potential delays and financial losses. Intellectual property (IP) protection concerns also remain a challenge, as companies may hesitate to share proprietary information with third-party developers. Additionally, limited capacity and resource constraints within contract development organizations can result in extended project timelines.
Opportunities
The growing pipeline of biologics, particularly in oncology, autoimmune diseases, and rare diseases, presents lucrative opportunities for contract development organizations. The increasing trend of small and mid-sized biotech firms partnering with CDOs to leverage their expertise and infrastructure further expands market opportunities. Additionally, advancements in single-use bioprocessing systems and continuous manufacturing technologies are streamlining biologics development, creating new growth avenues. Expansion into emerging markets, where governments are encouraging local biologics manufacturing and development, also provides opportunities for CDOs to establish a strong regional presence.
Challenges
A key challenge in the biologics contract development market is the need for stringent regulatory compliance across different regions. Navigating the complex and evolving regulatory landscape can be difficult for CDOs operating globally. Additionally, maintaining consistent product quality and ensuring supply chain reliability are critical concerns in biologics manufacturing. Talent shortages in specialized areas, such as cell line development and bioprocess optimization, pose another challenge for the market. Intense competition among CDOs and the growing trend of pharmaceutical companies developing in-house capabilities may further limit market growth.
Biologics Contract Development Market: Report Scope
| Report Attributes | Report Details |
|---|---|
| Report Name | Biologics Contract Development Market Research Report |
| Market Size in 2023 | USD 7.96 Billion |
| Market Forecast in 2032 | USD 16.18 Billion |
| Growth Rate | CAGR of 8.20% |
| Number of Pages | 196 |
| Key Companies Covered | WuXi Biologics, AGC Biologics, Genscript Biotech Corporation, Abzena Ltd., SE Thermo Fisher (Patheon), Bionova Scientific, LakePharma, STC Biologics, Inc., Hansen, Fujifilm Diosynth Biotechnologies, KBI Biopharma, Boehringer Ingelheim BioXcellence, and AGC Biologics |
| Segments Covered | By Source, By Product, By Disease and By Region |
| Regions Covered | North America, Europe, Asia Pacific (APAC), Latin America, Middle East, and Africa (MEA) |
| Base Year | 2023 |
| Historical Year | 2018 to 2022 |
| Forecast Year | 2024 - 2032 |
| Customization Scope | Avail customized purchase options to meet your exact research needs. Request For Customization |
Global Biologics Contract Development Market: Segmentation
The global biologics contract development market can be segmented into the source, product, disease, and region. All the segments have been analyzed based on present and future trends and the market is estimated from 2024 to 2032.
By source, the market can be segmented into mammalian, microbial, and others.
- The mammalian segment has the largest market share and is forecasted to witness significant growth rate in the coming years. The growth can be attributed to the usage of mammalian cells for protein synthesis to produce accurate post-transnational mammalian protein. Chinese mammalian cells are preferred the most for protein therapeutics and development for CMO cells.
- The microbial cell segment is expected to witness a high growth rate over forecast years. The microbial cell lines are majorly used in the production of hormones, enzymes, cytokines, and others.
By product, the market can be segmented into cell line development and process development.
The process development segment holds the largest share in the global biologics contract development market. The process development can further be segmented into upstream, downstream, and others.The upstream process includes the choosing of the right cell line, bioreactor system, operating dimensions, and culture medium.The downstream process includes the testing and purification of a fermented medium having an inoculum. Many qualitative assessments like pharmaceutical analysis, physicochemical characterization, packaging, and validating polishing are included in the downstream process segment.
By disease, the market can be segmented into oncology, immunological disorders, cardiovascular disorders, hematological disorders, and others.The oncology segment holds the largest market share.The immunology segment is expected to witness high growth rate over the forecast years. There are growing cases of immune system dysfunction all over the world.
The regional segment includes the current and forecast demand for North America, Europe, Asia Pacific, Latin America, and the Middle East and Africa.
Global Biologics Contract Development Market: Regional analysis
The Biologics Contract Development Market is experiencing dynamic growth across various regions, influenced by factors such as technological advancements, regulatory environments, and strategic investments. Here's a regional analysis highlighting key developments:
North America
North America remains a dominant force in the biologics contract development sector, with the United States leading due to its substantial consumer base and well-established providers. The U.S. market benefits from strong end-user demand and a focus on new product launches. The anticipated economic growth in 2024 is expected to further drive demand across various market segments.
Europe
Europe's biologics contract development market is characterized by proactive investment strategies aimed at understanding consumers, categories, competitors, and capabilities. Germany stands out as a significant market, with steady demand anticipated from countries like France, the UK, Spain, and Italy. Companies in this region are emphasizing effective marketing strategies and innovative product offerings to maintain a competitive edge.
Asia-Pacific
The Asia-Pacific region is emerging as a hub for biologics contract development, driven by increasing prevalence of various indications, robust healthcare expenditure, and significant investments in healthcare infrastructure. Countries such as China, India, and those in Southeast Asia present compelling growth opportunities. India, in particular, is witnessing substantial developments.
-
In November 2023, Aragen Life Sciences invested approximately $30 million to construct its first biologics manufacturing facility in India, aiming to meet the growing demand for biopharmaceuticals.
-
In March 2025, Syngene International acquired its first U.S. biologics facility for $36.5 million, enhancing its global footprint and capabilities in large molecule discovery, development, and manufacturing services.
Additionally, Indian contract drug manufacturers are seeking government support to reduce regulatory obstacles and expedite approval processes, aiming to strengthen their position in the global market.
Rest of the World
In regions such as Latin America, the Middle East, and Africa, continued urbanization and rising income levels are contributing to greater purchasing power and increased demand for biologics contract development services. Countries in the Middle East, including Saudi Arabia, the UAE, Qatar, and Kuwait, are experiencing robust demand, fueled by increasing healthcare expenditure and a growing population.
Global Biologics Contract Development Market: Competitive Players
Some of the prominent players in the global biologics contract development market are:
- WuXi Biologics
- AGC Biologics
- Genscript Biotech Corporation
- Abzena Ltd.
- SE Thermo Fisher (Patheon)
- Bionova Scientific
- LakePharma
- STC Biologics, Inc.
- Hansen
- Fujifilm Diosynth Biotechnologies
- KBI Biopharma
- Boehringer Ingelheim BioXcellence
- AGC Biologics.
The Global Biologics Contract Development Market is segmented as follows:
By Source
- Mammalian
- Microbial
- And Others
By Product
- Cell Line Development
- Process Development
By Disease
- Oncology
- Immunological Disorders
- Cardiovascular Disorders
- Hematological Disorders
- And Others
Global Biologics Contract Development Market: Regional Segment Analysis
- North America
- The U.S.
- Canada
- Europe
- France
- The UK
- Spain
- Germany
- Italy
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- Southeast Asia
- Rest of Asia Pacific
- Latin America
- Brazil
- Mexico
- Rest of Latin America
- Middle East & Africa
- GCC
- South Africa
- Rest of Middle East & Africa
What Reports Provides
- Full in-depth analysis of the parent market
- Important changes in market dynamics
- Segmentation details of the market
- Former, on-going, and projected market analysis in terms of volume and value
- Assessment of niche industry developments
- Market share analysis
- Key strategies of major players
- Emerging segments and regional markets
- Testimonials to companies in order to fortify their foothold in the market.
Table Of Content
FrequentlyAsked Questions
Biologics contract development is the comprehensive outsourcing service that manufactures biotechnology, pharmaceutical, and medical devices supporting biomaterials under the label of another company.
The Biologics Contract Development Market was valued at USD 7.96 Billion in 2023.
The Biologics Contract Development Market is expected to reach USD 16.18 Billion by 2032, growing at a CAGR of of 8.20% between 2024 to 2032.
Some of the significant factors that boost the global biologics contract development market are the surge in the adoption of advanced techniques for biological production, outsourcing the research & development tasks by pharma companies, and a well-suited environment for performing clinical trials. The surge in merger and collaboration activities increases the outsourcing of pharma and biopharma companies to expand in the developed and developing countries.
HappyClients
Zion Market Research
Tel: +1 (302) 444-0166
USA/Canada Toll Free No.+1 (855) 465-4651
3rd Floor,
Mrunal Paradise, Opp Maharaja Hotel,
Pimple Gurav, Pune 411061,
Maharashtra, India
Phone No +91 7768 006 007, +91 7768 006 008
US OFFICE NO +1 (302) 444-0166
US/CAN TOLL FREE +1 (855) 465-4651
Email: sales@zionmarketresearch.com
We have secured system to process your transaction.
Our support available to help you 24 hours a day, five days a week.
Monday - Friday: 9AM - 6PM
Saturday - Sunday: Closed